Hollow ShaftTM Sampling Method for Dissolution Testing
Richard Hengst and Rolf Rolli, Sotax Corporation, Furlong, PA
Introduction
With the increased testing demand in the laboratories of pharmaceutical companies, governmental institutions and contract service companies, automation becomes an important issue. Automated procedures can guarantee a higher throughput of samples with increased accuracy and a decreased variation due to human interference. With pressure to reduce the average cost per analysis automation is a viable solution. This paper compares Hollow ShaftTM sampling versus manual sampling in apparatus 1 and 2 dissolution testing.
Background
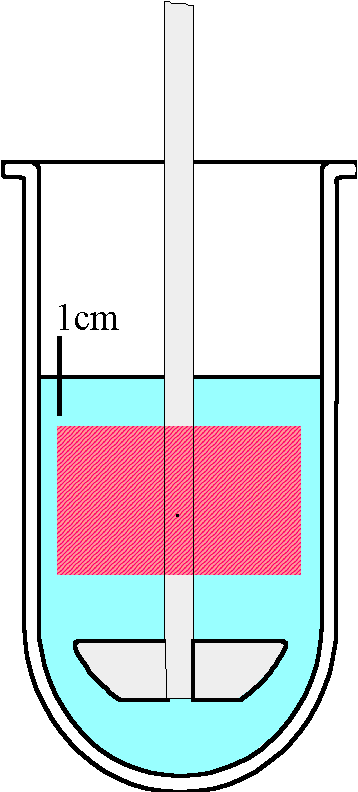
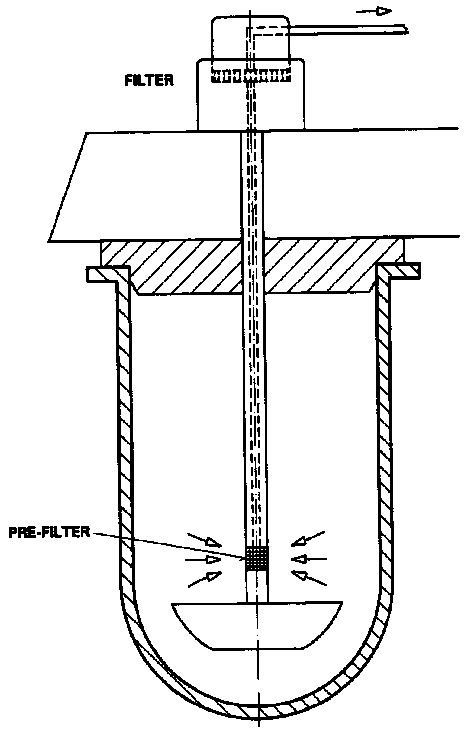
Sampling procedures in dissolution testing apparatus generate a great interest in the scientific community as they can influence test results significantly. Therefore, the regulatory authorities have taken this into consideration and have issued guidelines. The USP 23 [1] describes exactly how samples should be taken; "Withdraw a specimen from a zone midway between the surface of the Dissolution Medium and the top of the rotating basket or blade, not less than 1 cm from the vessel wall" (see figure 1). With the increased tendency to automate dissolution procedures this becomes even more important. The USP 23 requires "No part of the assembly, including the environment in which the assembly is placed, contributes significant motion, agitation, or vibration beyond that due to the smoothly rotating stirring element [1]. In USP apparatus 1 and 2 dissolution tests the samples are traditionally taken with a probe placed into the vessel. The probe adds turbulence if it remains in the liquid phase. Lift devices can be installed to move the probes in and out of the vessel at the required sampling times. In addition filtration with a probe sampling system may not meet the lab requirements. To overcome these problems, the Hollow ShaftTM sampling system was introduced several years ago and installed in numerous pharmaceutical laboratories throughout the world (figure 2). The Hollow ShaftTM sampling system allows for short sampling intervals, down to 20 seconds per sample and therefore the analysis of fast release drugs is possible.
 |
 |
| Figure 1: Pink rectangle is the sampling zone according to USP 23 | Figure 2: Hollow Shaft Sampling System |
The scientific community represented by the International Pharmaceutical Federation (FIP) issued new guidelines to reflect recent developments in testing, methodology and equipment [2]. In regards to automation, the guidelines state, "Validation of automated systems, concerning the sampling and analytical procedure, including media preparation and test performance, has to consider accuracy, precision and avoid contamination by any dilutions, transfers, cleaning or sample or solvent preparation procedures." It should be proven that there is no significant difference between data obtained with the manual dissolution equipment and the automated system, including manipulations such as permanent sampling probes, additional valves, hollow shaft, etc. In addition, the USP has issued guidelines regarding automated sampling and states[3] "If automated equipment is used for sampling and the apparatus is modified, validation of modified apparatus is needed to show that there is no change .."
Comparisons between manual and Hollow ShaftTM
sampling have already been presented to show that no significant
differences exist between the results of samples taken manually
or by means of Hollow ShaftTM [4,5]. This
study presents an extended comparison of manual (probe) sampling
versus Hollow ShaftTM sampling using semi
automatic on-line and off-line systems and a fully automated dissolution
system, the SOTAX AT70. The official USP calibrator tablets were
used as the test samples. All results have been taken from dissolution
systems located in pharmaceutical labs to show a realistic comparison
of equipment in routine use at different locations around the
world.
Equipment
The following equipment was used in the study:
 On-line: SOTAX AT7 dissolution tester with Hollow
ShaftTM sampling system and CY7-50 piston
pump connected to a Perkin Elmer Lambda 20 spectrophotometer
On-line: SOTAX AT7 dissolution tester with Hollow
ShaftTM sampling system and CY7-50 piston
pump connected to a Perkin Elmer Lambda 20 spectrophotometer
Off-line: SOTAX AT7 dissolution tester with Hollow ShaftTM sampling system and CY7-50 piston pump connected to a C 613 fraction collector
Filters: Whatman GF/D 2.7 µm microfiber filters
Fully automated system :
SOTAX AT70 with Hollow ShaftTM sampling system and CY7-50 piston pump connected to a Perkin Elmer Lambda 20 and/or to a fraction collector C 613 for combined on/off-line operation
Filters: Gelman 0.45 µm glass fiber filters
Sampling:
In all equipment, manual sampling through the SOTAX pipette guide ensured that the manual sample was consistently taken from the same position. The automated sampling was done through the Hollow ShaftTM.
Experimental Methods
All tests were executed according to the USP apparatus calibrator test using current lots of USP tablets and standards.
The dissolution medium was prepared with deionized water and the specified chemicals. The medium was deareated by helium sparging for 30 sec while maintaining a temperature of 40°C. After the test of 30 minutes the samples were withdrawn simultaneously. All samples have been measured with a Perkin-Elmer Lambda spectrophotometer except on the AT70 #97.1.001 where a Shimadzu UV-1601 was used. The spectrophotometers were equipped with either 1mm or 10mm flow through cuvettes . Standard readings were done before and after the sample measurements. The mean of both standard values was used to calculate % dissolved.
Test conditions
| Salicylic acid | Prednisone | |
| Standard: | Lot I, Weigh approx. 50mg and dissolve in 1000ml phosphate buffer pH 7.4 +/- 0,05 (to increase solubility 1% Ethanol was added) | Lot K, Weigh approx. 50mg and dissolve in 1000ml deionized water (to increase solubility 2% Ethanol was added) |
| Tablets: | Lot N, 300mg active substance | Lot L, 50mg active substance |
| Dissolution Medium: | 900ml deaerated phosphate buffer pH 7.40 +/- 0.05, 37.0 +/- 0.3°C | 900ml deaerated water, 37.0 +/- 0.3°C |
| Sampling Interval: | 30 minutes | 30 minutes |
| Measurement: | 296nm | 242nm |
| Paddle: | 100 rpm | 50 rpm |
| Basket: | 100 rpm | 100 rpm |
Results
The apparatus involved in the study are described by the serial numbers. The sampling method is coded as follows:
M: Manual
HS on-line: Hollow shaft sampling on-line with spectrophotometer (see Equipment).
HS off-line: Hollow shaft sampling off-line with fraction collector and subsequent measurement with spectrophotometer (see Equipment).
The % dissolved results from each vessel, the mean and the difference of manual sampling minus hollow shaft sampling is shown in figure 3.
Figure 3.
AT7 on-line systems
The following systems were used in the study: AT7 #96.4.162 , AT7 #96.4.145.
AT7 off-line systems
The following systems were used in the study: AT7 #91.1.074 two studies, AT7 #91.1.056, AT7 #94.4.098, AT7 #94.4.097, AT7 #91.1.056.
AT70 on-line and combined on/off-line
The following systems were used in the study: AT70 #97.1.001,
AT70 #97.1.002, AT70 #97.1.005, AT70 #97.1.006, AT70 #97.1.007,
AT70 #97.1.008
All data for the paddle method were combined to recalculate mean and standard deviation to have a clear statistical comparison.


Conclusion
The data show that samples taken by means of the Hollow ShaftTM method are comparable to the results taken by a manual method. The system of Hollow ShaftTM sampling is fully compliant with all current FIP,USP and FDA guidelines and is an efficient sampling method. The Hollow ShaftTM sampling system can be utilized in a range of configurations from fully automated to on- or off-line operation for reliable and consistent data.
References
[1] USP 23, January 1, 1995 page 1791
[2] Pharmacopeial Forum, Volume 21, Number 5, 1371-1382
[3] Pharmacopeial Forum 23 Number 6 (Nov-Dec 1997), page 5257
[4] Dissolution Technologies, Vol. 3, issue 2, May 1996, 11-15
[5] Rolf Rolli oral presentation at FIP Spring Meeting, 15th May 1997, Allschwil