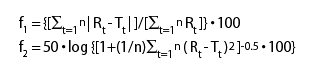
Dissolution
Profile Comparison Using
Similarity Factor, f2
Vinod P. Shah, Yi Tsong, Pradeep
Sathe and Roger L. Williams
Office of Pharmaceutical
Science, Center for Drug Evaluation and Research
Food and Drug Administration, Rockville, MD
What is a dissolution profile comparison and what is its impact?
In recent years, FDA has placed more
emphasis on a dissolution profile comparison in the area of post-approval
changes and biowaivers. Under appropriate test conditions, a dissolution
profile can characterize the product more precisely than a single
point dissolution test. A dissolution profile comparison between
pre-change and post-change products for SUPAC related changes,
or with different strengths, helps assure similarity in product
performance and signals bioinequivalence.
Among several methods investigated for dissolution profile comparison, f2 is the simplest. Moore and Flanner proposed a model independent mathematical approach to compare the dissolution profile using two factors, f1 and f2 (1).
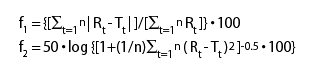
where Rt and Tt are the cumulative
percentage dissolved at each of the selected n time points of
the reference and test product respectively. The factor f1 is
proportional to the average difference between the two profiles,
where as factor f2 is inversely proportional to the average squared
difference between the two profiles, with emphasis on the larger
difference among all the time-points. The factor f2 measures the
closeness between the two profiles. Because of the nature of measurement,
f1 was described as difference factor, and f2 as similarity factor
(2). In dissolution profile comparisons, especially to assure
similarity in product performance, regulatory interest is in knowing
how similar the two curves are, and to have a measure which is
more sensitive to large differences at any particular time point.
For this reason, the f2 comparison has been the focus in Agency
guidances.
When the two profiles are identical,
f2=100. An average difference of 10% at all measured time points
results in a f2 value of 50. FDA has set a public standard of
f2 value between 50-100 to indicate similarity between two dissolution
profiles.
For a dissolution profile comparison:
A general question is how large can the difference between the mean dissolution profiles be before the difference is likely to impact on in vivo performance. From a public health point of view, and as a regulatory consideration, a conservative approach is appropriate. The f2 comparison metric with a value of 50 or greater is a conservative, but a reliable estimate to assure product sameness and product performance.
References:
1. J.W.Moore and H.H.Flanner, Mathematical Comparison of curves
with an emphasis on in vitro dissolution profiles. Pharm. Tech.
20(6), : 64-74, 1996.
2. V.P.Shah, Y.Tsong and P.Sathe, In vitro dissolution profile comparison - statistics and analysis of the similarity factor, f2. Pharm. Res. 15: 889-896, 1998.